rpc00008: Arabidopsis thaliana T87 cell suspension culture
Note
Components
Domestic delivery: A 50-mL plastic conical centrifuge tube, containing cell suspension
Overseas delivery: A 250-mL plastic Erlenmeyer flask, containing cells placed on semi-solid medium
Notice
Subculture the cells to fresh medium immediately after arrival.
Do not store the cell culture in a refrigerator and a freezer.
Maintain aseptic conditions of the cell culture, and work in a laminar flow cabinet.
Method
Culture medium: JPL medium, 1 µM NAA %, pH %5.7 (medium no. 5)
Culture conditions: 22°C, continuous light, 120 rpm
Subculture: 14-day intervals
Citation of cell line
When results obtained by using this cell line are published in a scientific journal, it should be cited in the following manner: “Arabidopsis thaliana T87 cell line (rpc00008) was provided by the RIKEN BRC through the National BioResource Project of the MEXT, Japan.”
Introduction
Arabidopsis T87 cell line was established from a seedling of Arabidopsis thaliana (L.) Heynh. accession Columbia (Axelos et al. 1992 [1]). The T87 cell culture is green and composed of small near-uniform aggregates of cells (Figure 1). The T87 cells are grown in a Jouanneau and Péaud-Lenoël (JPL) medium supplemented with 1 µM 1-naphthaleneacetic acid (NAA). Our T87 cell culture has been maintained under the continuous light at 22°C with rotary shaking at 120 rpm and subcultured at 14-day intervals (Figure 2).
Materials
Chemicals and stock solutions
(All stock solutions are stored at 4°C)
JPL_Aʼ
Chemical
Concentration (mg/mL)
KNO3
65.5
CaCl2·2H2O
4.4
MgSO4·7H2O
3.7
KH2PO4
1.7
JPL_B
Chemical
Concentration (mg/mL)
H3BO3
6.2
MnSO4·5H2O
24.1
ZnSO4·7H2O
10.6
KI
0.83
Na2MoO4·2H2O
0.25
CuSO4·5H2O
0.025
CoCl2·6H2O
0.025
JPL_C
Chemical
Concentration (mg/mL)
FeSO4·7H2O
2.78
Na2-EDTA
3.73
Heat at 80°C for 3–4 hours for chelating Fe
JPL_D
Chemical
Concentration (mg/mL)
Glycine
0.2
myo-Inositol
10
JPL_VT
Chemical
Concentration (mg/mL)
Nicotinic acid
0.5
Pyridoxine·HCl
0.5
Thiamine·HCl
0.4
JPL_P
Stock solution
Volume (mL)
200 mM KH2PO4
19.5
200 mM Na2HPO4
30.5
H2O
50
NAA (1 mM)
Chemical
Concentration (mg/mL)
NAA·K
0.224
Potassium 1-naphthylacetate, FUJIFILM Wako Pure Chemical Corporation (#161-04021)
Sucrose
Casein hydrolysate, vitamin free
Casamino acids vitamin assay, Difco (#228820)
KOH (1 N)
HCl (1 N)
Glassware and equipment
(All are sterilized by autoclaving at 121°C for 20 min)
Erlenmeyer flask (300 mL), capped with two layers of aluminum foil
Pipette (10 mL; large tip opening) and a bulb
Stainless sieve (diameter, 5 cm; pore size, 1 mm) set on a tall beaker (200 mL), capped with two layers of aluminum foil
Preparation of JPL medium (medium no. 5)
Prepare three solutions as follows.
JPL mineral solution (1 L)
Stock solution
Volume (mL)
JPL_Aʼ
37.5
JPL_B
0.375
JPL_C
2.5
Adjust the pH to 5.7 with KOH (1 N)
JPL organic solution (100 mL)
Chemical/Stock solution
Amount
JPL_D
10 mL
JPL_VT
1 mL
Casein hydrolysate
0.1 g
Adjust the pH to 5.7 with HCl (1 N)
JPL sucrose solution (100 mL)
Chemical/Stock solution
Amount
JPL_P
1 mL
NAA (1 mM)
1 mL
Sucrose
15 g
Autoclave the solutions at 121°C for 20 min.
Add 800 mL of JPL mineral solution, 100 mL of JPL organic solution, and 100 mL of JPL sucrose solution asceptically.
Pour 80 mL of the medium into a sterile 300-mL flask.
Methods
Filter a 14-day-old cell suspension through a stainless sieve (Figure 3).
Agitate the filtrate well and transfer 2–3 mL of cell suspension to 80 mL of fresh JPL medium with a pipette.
Incubate cell cultures on a rotary shaker at 120 rpm under the continuous light condition (photosynthetic photon flux density 40–42 µmol m-2 s-1) at 22°C.
Notes
For domestic customers: We send T87 cell suspension in a 50-mL disposable conical centrifuge tube. The cells should be transferred to fresh JPL medium immediately after arrival.
For overseas customers: We send T87 cells placed on semi-solid JPL medium in a 250-mL disposable Erlenmeyer flask. The cells should be transferred to fresh JPL medium immediately after arrival. Collect the cells from the semi-solid medium with a spatula and transfer them to Erlenmeyer flasks containing fresh liquid medium.
In order to maintain T87 cell suspension cultures stably, it is essential to transfer an adequate amount of cells to fresh JPL medium in every subculture. The amount of cells may vary from one lab to another, because proliferation of T87 cells is affected by culture conditions, such as a room temperature, rotation speed of a rotary shaker, and aeration condition of the culture. T87-cell clumps occasionally develop into large aggregates, which causes a decrease in the amount of cells passed through a 1-mm sieve.
In order to obtain good aeration of a suspension culture, a silicone sponge plug may be used instead of the aluminum foil cap (e.g., cap-type Silicosen; Shin-Etsu Polymer, Tokyo, Japan).
T87 cells may be maintained in Gamborgʼs B5 medium (Yamada et al. 2004 [3]) or Murashige and Skoog medium (Cunillera et al. 2000 [2]) other than JPL medium.
References
Genotyping
Maintenance history
Protocols
Cryopreservation of Arabidopsis T87 suspension cell cultures
Genotyping of plant cell lines: Arabidopsis thaliana cell lines
External links
Figures
Figure 1

Arabidopsis thaliana T87 cell suspension culture
Two-week-old cell suspension culture.
Cell clumps. The size of cell clumps are varied, but most of them are below 1 mm. Scale bar = 1 mm
Morphology of T87 cells. T87 cells were observed by using differential interference contrast microscopy. The cells contain many chloroplasts. Scale bar = 50 µm
Figure 2
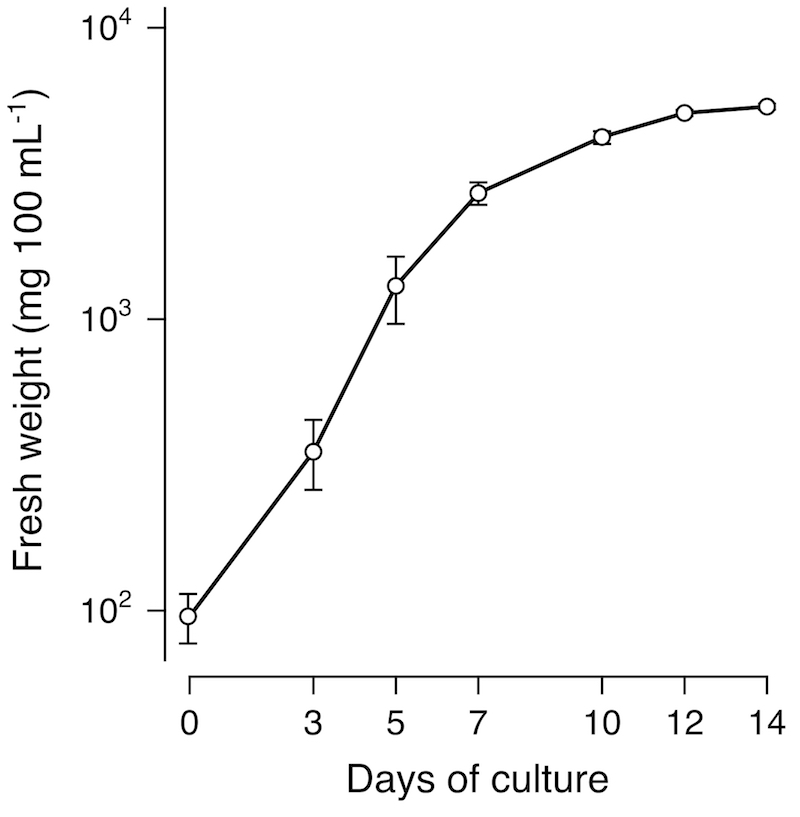
Growth profile of T87 cells
Figure 3

Procedure for subculturing T87 cells
Two-week-old cell suspension culture.
Pass the culture through a stainless sieve (pore size, 1 mm).
Transfer 2 mL of the filtrate to 80 mL of fresh JPL medium.